
Chromium metal can be plated out from an acidic solution containing CrO3 according to the following equation CrO3(aq) + 6H^ + (aq) + 6e^ - → Cr(s) + 3H2O Calculate how many

Measuring the thickness of the silver layer when electroplating plates for daguerreotype — Absolutely Analog












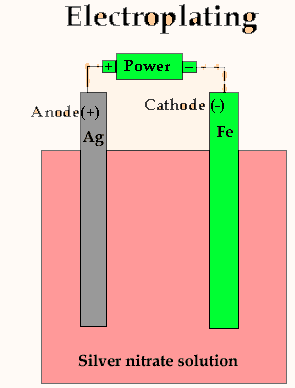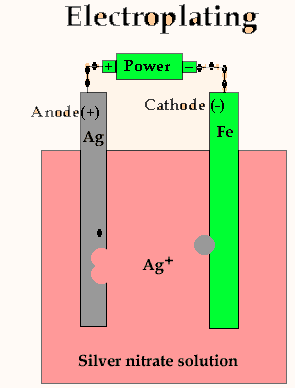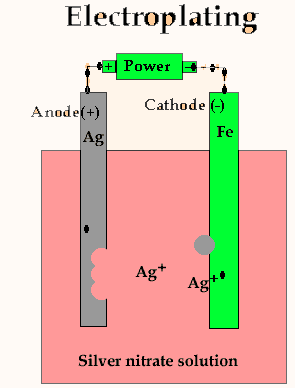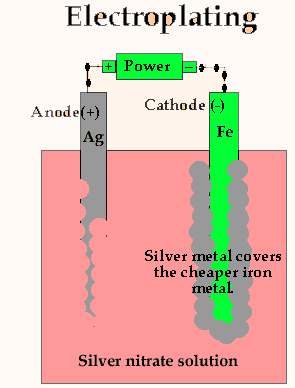

![Electroplating [SubsTech] Electroplating [SubsTech]](https://www.substech.com/dokuwiki/lib/exe/fetch.php?w=&h=&cache=cache&media=electroplating.png)


